
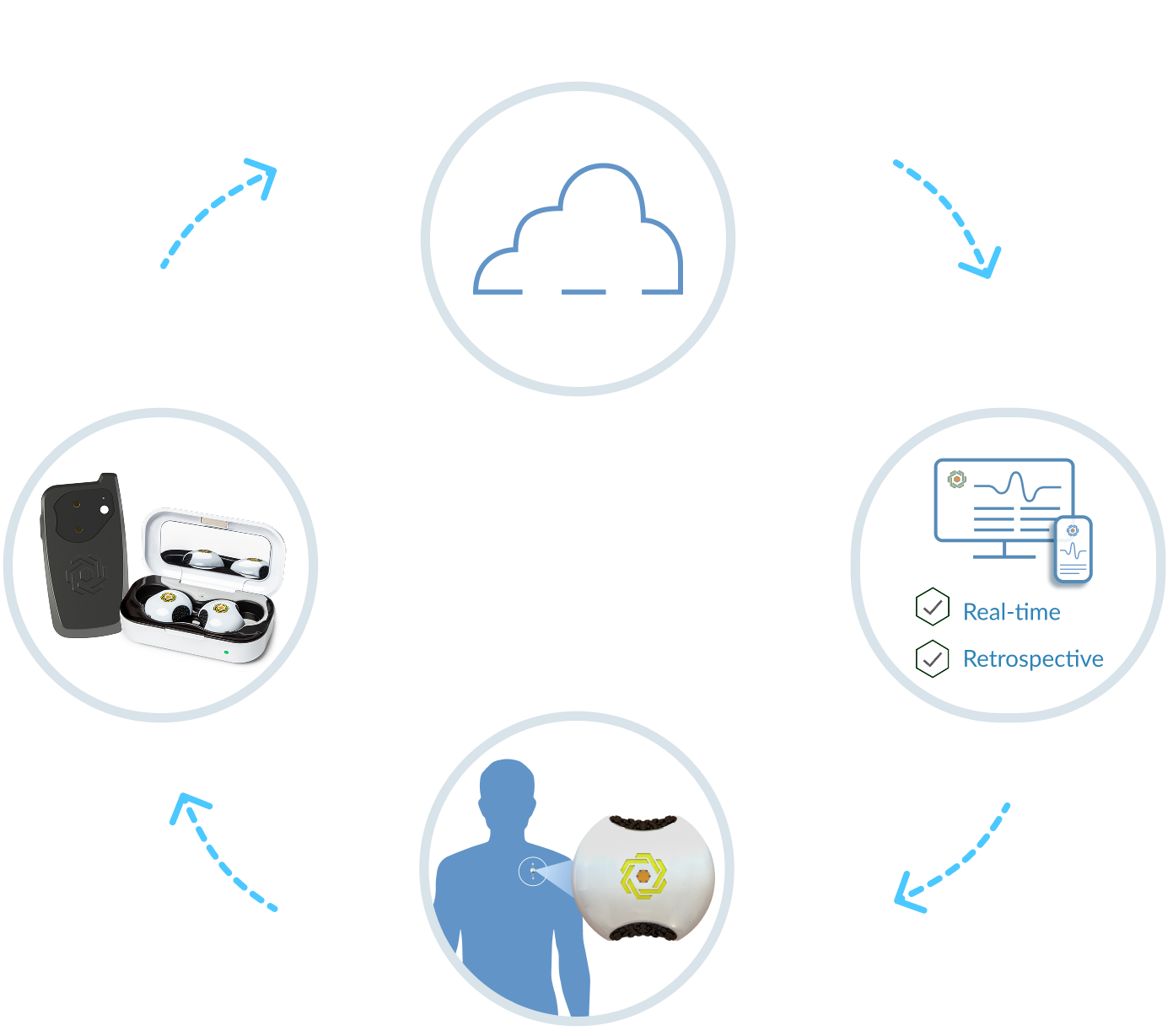
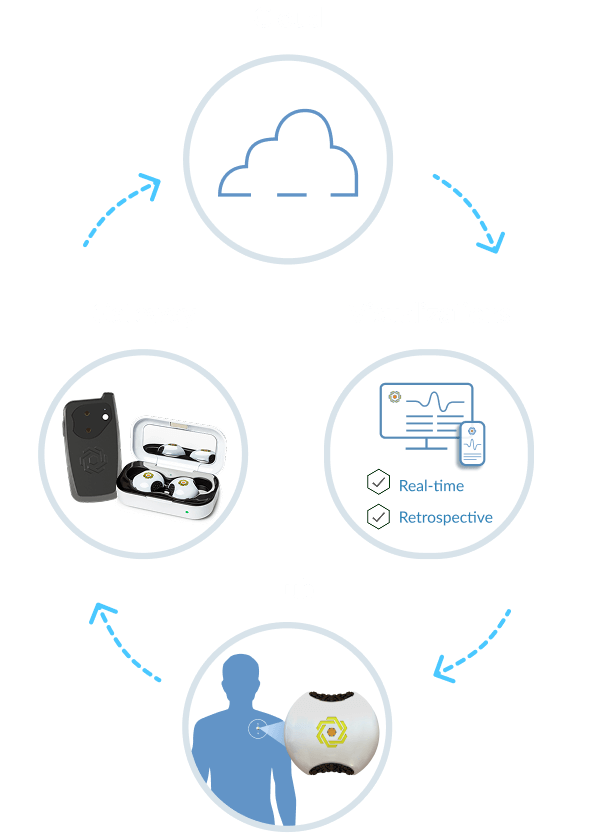
By gathering all the data needed, all at once, all in real-time, the LifeLens Physiological Monitoring Platform provides the big picture required to achieve a more complete understanding of an individual’s health.
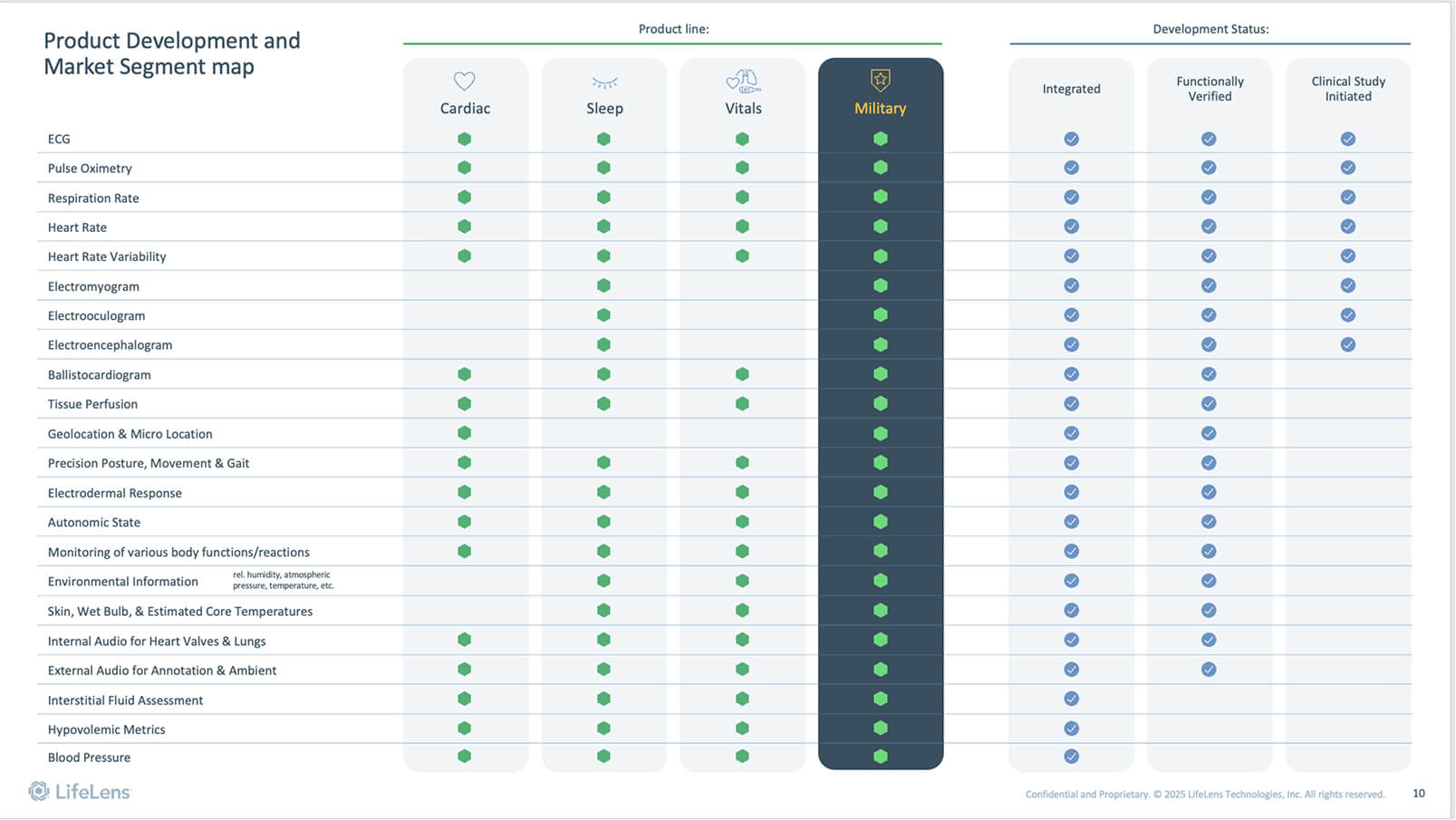
The technology that drives the comprehensive monitoring platform of LifeLens has been tested by the U.S. military in over 40 fielding events since 2021. During this time, it has helped to monitor clinical-grade vital signs, mitigate heat and musculoskeletal injuries, and determine unit readiness.
And this is only the beginning.
+
LifeLens® Wireless ECG Monitor is indicated for use on patients who maybe asymptomatic or who may suffer from transient symptoms such as palpitations, shortness of breath, dizziness, light headedness, pre-syncope, syncope, fatigue, chest pain and/or anxiety. The LifeLens® Wireless ECG Monitor is intended for use by patients 18 years or older. The LifeLens® Wireless ECG Monitor is not intended to be worn during defibrillation.
PRIVACY POLICY
LifeLens® word and logo mark are registered trademarks of LifeLens® Technologies, Inc. in the United States. LifeLens® Wireless ECG Monitor is indicated for use on patients who may be asymptomatic or who may suffer from transient symptoms such as palpitations , shortness of breath, dizziness, light headedness, pre-syncope, syncope, fatigue, chest pain and / or anxiety. The LifeLens® Wireless ECG Monitor is intended for use by patients 18 years or older. The LifeLens® Wireless ECG Monitor is not intended to be worn during defibrillation. The system is not yet FDA cleared for other indications.
