
Wearable devices serve as a bridge between technology and human performance by providing objective physiological data for assessing warfighter readiness, health, and performance. Given their innovative nature and application in military operational settings, feedback from end users is crucial for determining the usability of wearable devices. Therefore, this study aimed to evaluate the usability and end-user comfort of the LifeLens device.

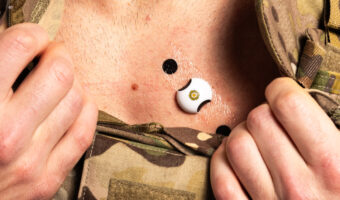
LifeLens® Wireless ECG Monitor is indicated for use on patients who maybe asymptomatic or who may suffer from transient symptoms such as palpitations, shortness of breath, dizziness, light headedness, pre-syncope, syncope, fatigue, chest pain and/or anxiety. The LifeLens® Wireless ECG Monitor is intended for use by patients 18 years or older. The LifeLens® Wireless ECG Monitor is not intended to be worn during defibrillation.
PRIVACY POLICY
LifeLens® word and logo mark are registered trademarks of LifeLens® Technologies, Inc. in the United States. LifeLens® Wireless ECG Monitor is indicated for use on patients who may be asymptomatic or who may suffer from transient symptoms such as palpitations , shortness of breath, dizziness, light headedness, pre-syncope, syncope, fatigue, chest pain and / or anxiety. The LifeLens® Wireless ECG Monitor is intended for use by patients 18 years or older. The LifeLens® Wireless ECG Monitor is not intended to be worn during defibrillation. The system is not yet FDA cleared for other indications.
