
Fort Carson, Colo. – The 4th Infantry Division tested advanced wearable sensors and end-user software during Ivy Sting 4, demonstrating how real-time physiological and environmental data can improve casualty triage, treatment, and tracking during simulated combat trauma, Jan. 26 to Feb. 5, 2026.
The technology supported sensor-assisted triage, treatment, and tracking (SAT3) across multiple brigades under realistic large-scale combat operations (LSCO) conditions.

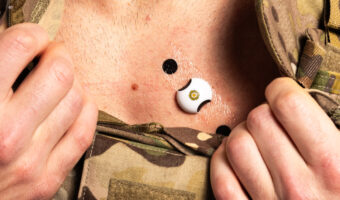
LifeLens® Wireless ECG Monitor is indicated for use on patients who maybe asymptomatic or who may suffer from transient symptoms such as palpitations, shortness of breath, dizziness, light headedness, pre-syncope, syncope, fatigue, chest pain and/or anxiety. The LifeLens® Wireless ECG Monitor is intended for use by patients 18 years or older. The LifeLens® Wireless ECG Monitor is not intended to be worn during defibrillation.
PRIVACY POLICY
LifeLens® word and logo mark are registered trademarks of LifeLens® Technologies, Inc. in the United States. LifeLens® Wireless ECG Monitor is indicated for use on patients who may be asymptomatic or who may suffer from transient symptoms such as palpitations , shortness of breath, dizziness, light headedness, pre-syncope, syncope, fatigue, chest pain and / or anxiety. The LifeLens® Wireless ECG Monitor is intended for use by patients 18 years or older. The LifeLens® Wireless ECG Monitor is not intended to be worn during defibrillation. The system is not yet FDA cleared for other indications.
